hidden title
Clinical Development Support Services
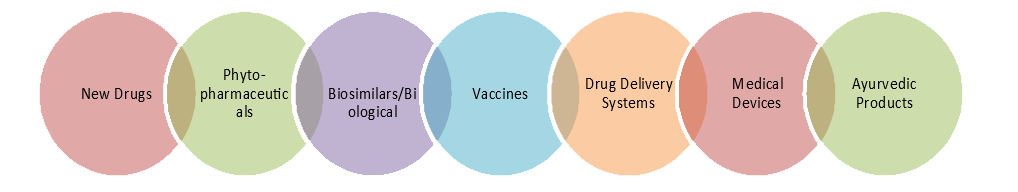
BCIL adopts an integrated approach for facilitating preclinical and clinical development (Phase-1 to IV) of healthcare innovations covering identification of jurisdictions with market potential and ensuring generation of regulatory data in conformity with the specific regulations for time and cost-effective clinical development of the product.
Services Offered
Identification of most suitable regulatory pathway for the new product taking into consideration
- Jurisdictions for marketing of the product
- Waiver/exemption available as per current regulations
- Minimum data compliance in view of approved products
- Filing of investigative new drug (IND) application with the relevant regulatory authorities
- Developing detailed plan for preclinical and clinical development and its implementation conforming strictly to the timelines
- End to end management of the study in a timeline-based manner for expedited product launch
BCIL develops a timeline-based implementation plan and ensures strict compliance with the timelines to ensure expediency of movement from one stage of Clinical Development to the next without loss of time and consequently prevent erosion of Patent Life.